
ACOUSTICS AND MICROFLUIDICS
Acoustophoresis: the art of combining acoustics and microfluidics
The manipulation of particles is an essential feature of Lab-on-a-chip (LOC) devices. LOC-systems provide a platform where biochemical assays can be miniaturized, integrated and automated in a simple, portable, fast and cost-efficient way [1].
Acoustophoretic devices are a type of LOC systems in which particles can be moved and spatially localized by sound. An acoustophoretic system is typically made of a chip with embedded micro channels and a piezoelectric transducer attached underneath, responsible for the ultrasound actuation [2].
If the frequency of the ultrasound actuation is properly tuned, a standing wave or resonance condition is established in the microfluidic channels.
Typically, the system is built such that a single pressure node is generated in the center of the flow channel. Suspended particles flowing in a standing wave field are affected by acoustic radiation forces, that move them into specific locations in the microchannel depending on physical properties (size, density and compressibility) of the particles compared to the surrounding medium [3-6] (Fig.1).
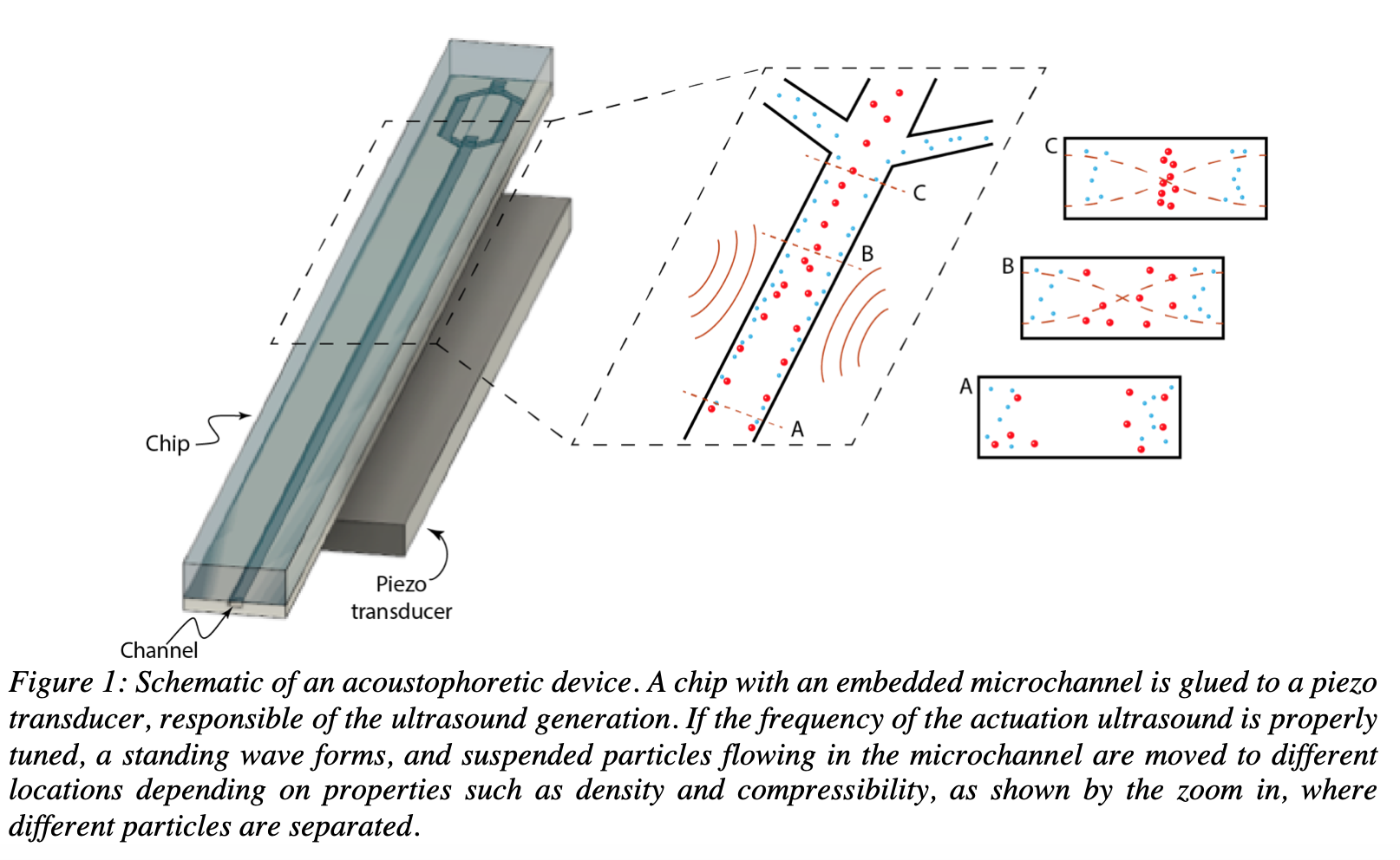
Commonly, solid particles such as cells or microbeads are moved into the pressure node, while softer particles, such as lipids and gas bubbles move towards the antinode.
Because of the laminar flow conditions in the microchannel, a particle holds its lateral position in the continuous flow even after passing through the sound field. A flow splitter enables a division of flow streams, thus allowing for particle fractionation based on their intrinsic acoustic properties.
This can be used for unit operations such as alignment [7], separation [8, 9], washing [10] and trapping [11] of particles and cells.
As these microfluidic devices work with very small sample volumes, they are ideal for biomedical sample preparations and separation and sorting of cell populations for clinical analysis. Within the BioWings project, new improved acoustophoresis chips will be developed that will target the unmet need for efficient microscale plasmapheresis and rapid bacteria separation for sepsis applications as well as rapid point-of-care hematocrit determination.
References
- Mark, D., et al., Microfluidic lab-on-a-chip platforms: requirements, characteristics and applications. Chemical Society Reviews, 2010. 39(3).
- Nilsson, A., et al., Acoustic control of suspended particles in micro fluidic chips. Lab Chip, 2004. 4(2): p. 131-5.
- King, L.V., On the acoustic radiation pressure on spheres. Proceedings of the Royal Society of London. Series A-Mathematical and Physical Sciences, 1934. 147(861): p. 212-240.
- Yosioka, K. and Y. Kawasima, Acoustic radiation pressure on a compressible sphere. Acta Acustica united with Acustica, 1955. 5(3): p. 167-173.
- Gor’kov, L.P. On the forces acting on a small particle in an acoustical field in an ideal fluid. in Sov. Phys. Dokl. 1962.
- Settnes, M. and H. Bruus, Forces acting on a small particle in an acoustical field in a viscous fluid. Physical Review E, 2012. 85(1): p. 016327.
- Goddard, G., et al., Ultrasonic particle‐concentration for sheathless focusing of particles for analysis in a flow cytometer. 2006. 69(2): p. 66-74.
- Petersson, F., et al., Separation of lipids from blood utilizing ultrasonic standing waves in microfluidic channels. Analyst, 2004. 129(10): p. 938-43.
- Augustsson, P., et al., Microfluidic, label-free enrichment of prostate cancer cells in blood based on acoustophoresis. Anal Chem, 2012. 84(18): p. 7954-62.
- Tenje, M., et al., Acoustophoretic removal of proteins from blood components. Biomed Microdevices, 2015. 17(5): p. 95.
- Evander, M. and J. Nilsson, Acoustofluidics 20: applications in acoustic trapping. Lab Chip, 2012. 12(22): p. 4667-76.






