
Demographic trends, such as the rapid growth and ageing of the world population, are putting pressure on global healthcare systems, increasing the demand for smart, effective and affordable biomedical systems. Micro-Electro- Mechanical Systems (MEMS) are key components of such biomedical systems, enabling miniaturised devices with diagnostic, prognostic and therapeutic functionalities.
Although these systems are poised to revolutionize medical diagnostics and treatment approaches, the slow progress in the development of biocompatible actuator materials is still hindering this industry, preventing a host of new biomedical devices to enter the mainstream market.
BioWings proposes to solve this deadlock through the implementation of a completely new class of smart actuating materials to be integrated in biocompatible MEMS. This family of materials is based on highly defective cerium oxides, which recently displayed radically different properties compared to existing ones:
- They are non-toxic and environmentally friendly, unlike the current lead-based actuators.
- They show exceptionally high and still uncapped electrostrictive response under moderate electric fields, enabling low power consumption devices.
- They are fully compatible with silicon-based technologies and many other substrates, including metals and polymers.
To fully explore the potential of these materials, foundational knowledge must still be generated on both the basic physical mechanisms and the manufacturing process. To reach this, BioWings focuses on:
- Understanding, predicting, and controlling the mechanism underlying the unparalleled electrostrictive behaviour of highly defective oxides, by unveiling the effects of the microstructure, as well as the type and concentration of dopants.
- Identifying a methodology for controlling the electromechanical properties of such materials, using facile manufacturing processes on bio-compatible substrates and electrodes, exploring the scale limit of the device/materials, thus opening up a new path that solves important manufacturing issues in advanced electronics industry.
- Proving the concept by integrating ceria-based electrostrictors into Bio-MEMS with diverse architectures and acoustofluidic medical blood samples preparation chips.
Such results will be pursued by a multidisciplinary group of academic, industrial and medical partners, who will lay the foundations for a new paradigm in a new bio-compatible and environmentally friendly actuator smart materials design and implementation, which will have considerable impact on the scientific, medical and industrial community.

The vision of artificial biomedical systems capable of monitoring, healing or even replacing damaged functionalities in the human body is emerging, pushed by the rapid development of miniaturized electronics and new materials.
Smart materials – suchas piezoelectric (PE) and electrostrictive (ES) materials – are contributing to this revolution, allowing previously unthinkable products and functionalities to be available in various application scenarios. These have already found their way into key components in consumer electronics, biomedical devices, robots, aerospace, wireless communication and automotive systems.
In order to fully realise the vision of bio-inspired systems, smart materials should be pushed far beyond their current capabilities, radically upgrading their performance in terms of displacement, force, bandwidth, fatigue resistance, and biocompatibility. Besides, smart materials of the future should be monolithically integrated into a wide range of substrates, including polymers, metals, glass, and silicon.
As muscles, electrostrictors (ES) are capable of changing shape and produce a mechanical stress in response to an electrical field. The recent discovery of giant ES in acceptor-doped ceria compounds thin films has attracted great interest for its outstanding properties, promoting this class of materials as an extremely promising candidate for the next generation of actuators, e.g. for moving parts and mechanical waves generators.
With this in mind, the ultimate scientific goal of the BioWings project is to master these material’s properties in order to integrate it in biocompatible technologies and to realize the next generation of smart materials for miniaturized medical devices. To achieve such goal and unlock a wide variety of medical future applications including microfluidic MEMS systems, simple moving parts for implantable pumps, valves, drug delivery systems, MEMS architectures will be prototyped and validated at laboratory stage (TRL4) under a wide range of operational frequencies and substrates.

BioWings will pursue the following main objectives:
- RESEARCH: Fundamental understanding of giant ES in ceria and biocompatibility
The objective is to explore the fundamental boundaries of the ES phenomenon in ceria, understanding the mechanism underlying it, and defining the methodology to design and implement actuators with the characteristics required by the specific applications. - IMPLEMENTATION: Designs and architectures (electrodes and substrates) that maximize the ES effect on bio substrates
The objective is to identify the optimal composition of the material for its integration in MEMS and other bio-substrates. The steps expected at this stage include materials engineering aspects of the electrostrictive materials based on doped ceria, as well as theoretical modelling and simulations of the ES- based actuators and of the ES-actuated microfluidic devices. - APPLICATIONS: use on devicesThe objective of this phase is to select possible designs for bio-med-MEMS for various applications both for the membrane design and the fully supported films by implementing the materials designed and tested. After defining the device portfolio, we will proceed with lab-scale prototyping in two medical applications: Performance in removing blood cells from the blood plasma and image analysis of focused blood cells in a microfluidic acoustic resonator, thus enabling accurate estimation of the haematocrit level (HCT) of a blood sample.
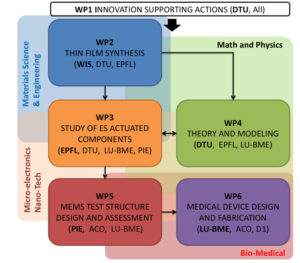
The project has a planned duration of 54 months, with a work plan subdivided in 6 work packages. The plan is structured as follows:
WP1 will deal with the project management.
WP2 we will carry out the synthesis and microstructural characterisation of the electrostrictive films, investigating the influence of deposition parameters, substrates, and electrodes on the film characteristics.
In parallel, WP3 will take care of mastering the giant ES effect in the thin films produced in WP2, by analysing the effect of dopants, microstructure, and defective crystals.
This work will be strongly connected with the modelling activities run in WP4, which will provide a tool for predicting the behaviour of the devices and the fluids under analysis.
Once the properties of the electrostrictive thin-film are known and optimized, WP5 will focus on the design, integration and characterisation of the first actuator modules on various substrates. Mechanical simulation will also be used to streamline and control the design process.
Finally, in WP6 the actuators will be integrated on a prototypal biomedical system.
 The project is expected to produce 41 deliverables out of the activities carried out in the 6 work packages.
The project is expected to produce 41 deliverables out of the activities carried out in the 6 work packages.
Most of them are considered confidential, since they will contain results produced during the project and likely to be exploited through technology transfer and/or spin-off activities.
This page contains the list of deliverables, and those that are public can be downloaded by clicking on the title.
D1.1 Dissemination and Communication Plan and Tools
D1.2 Dissemination and Communication Plan and Tools 2
D1.3 Dissemination and Communication Plan and Tools 3
D1.4 IPR Protection and Exploitation Plan
D1.5 IPR Protection and Exploitation Plan 2
D1.6 End-users’ requirements and technical specs
D1.7 White Paper for Investors
D1.8 Project logo and website
D1.9 Open Access Scientific Publications
D1.10 Data Management Plan
D1.11 Technical/scientific review meeting documents
D1.12 Technical/scientific review meeting documents – 2
D1.13 Technical/scientific review meeting documents – 3
D2.1 Thin films for ES measurements
D2.2 Thin films for ES measurements 2
D2.3 Thin films for ES measurements 3
D2.4 Multilayer deposition methods and substrates
D2.5 Optimal deposition methodologies
D2.6 Optimized BioWings defined
D3.1 Parameters for ES maximization
D3.2 Optimal material microstructure
D3.3 Effect of dopant on ES performance optimization
D3.4 In-situ measurement methodology for device testing
D4.1 First models ofthe ES-actuator membrane pump and acoustofluidic device for WP6
D4.2 Final models of ES- actuators on a solid support
D4.3 Final model of the ES- actuated ultrasound acoustofluidic devices
D4.4 Final model of the membrane actuator for optimized actuator design+
D5.1 Designs and specifications of the actuator based on the optimized films
D5.2 Designs and specifications of the actuator based on the optimized films 2
D5.3 Designs and specifications of the actuator based on the optimized films 3
D5.4 Electromechanical charact. of the membrane actuator
D5.5 Manufacturing and testing of the first high frequency ceria-ES actuated acoustophoresis chips
D5.6 Actuators designs delivered
D5.7 MEMS fabricated
D5.8 Long term performance evaluation
D5.9 Actuators on polymer feasibility and fabr. method
D6.1 HTC and sepsis acoustofluidic chips on PZT
D6.2 HTC and sepsis protocols in clinical samples on PZT
D6.3 Ceria-ES integrated acoustofluidic chips
D6.4 Performance of Ceria-ES acoustofluidic devices
D6.5 Application areas for the new ES-actuator material
DISSEMINATION MATERIALS
Whether you are a scientific magazine, a journalist with an editorial focus on new technologies, a researcher, or a simple innovation enthusiast, in this section you can find the communication material for the BioWings project.
Discover, share, print!
The BioWings Brochure – Page 1
Click the icon to download


